Disruptive next generation pneumococcal vaccine
Disruptive next generation pneumococcal vaccine
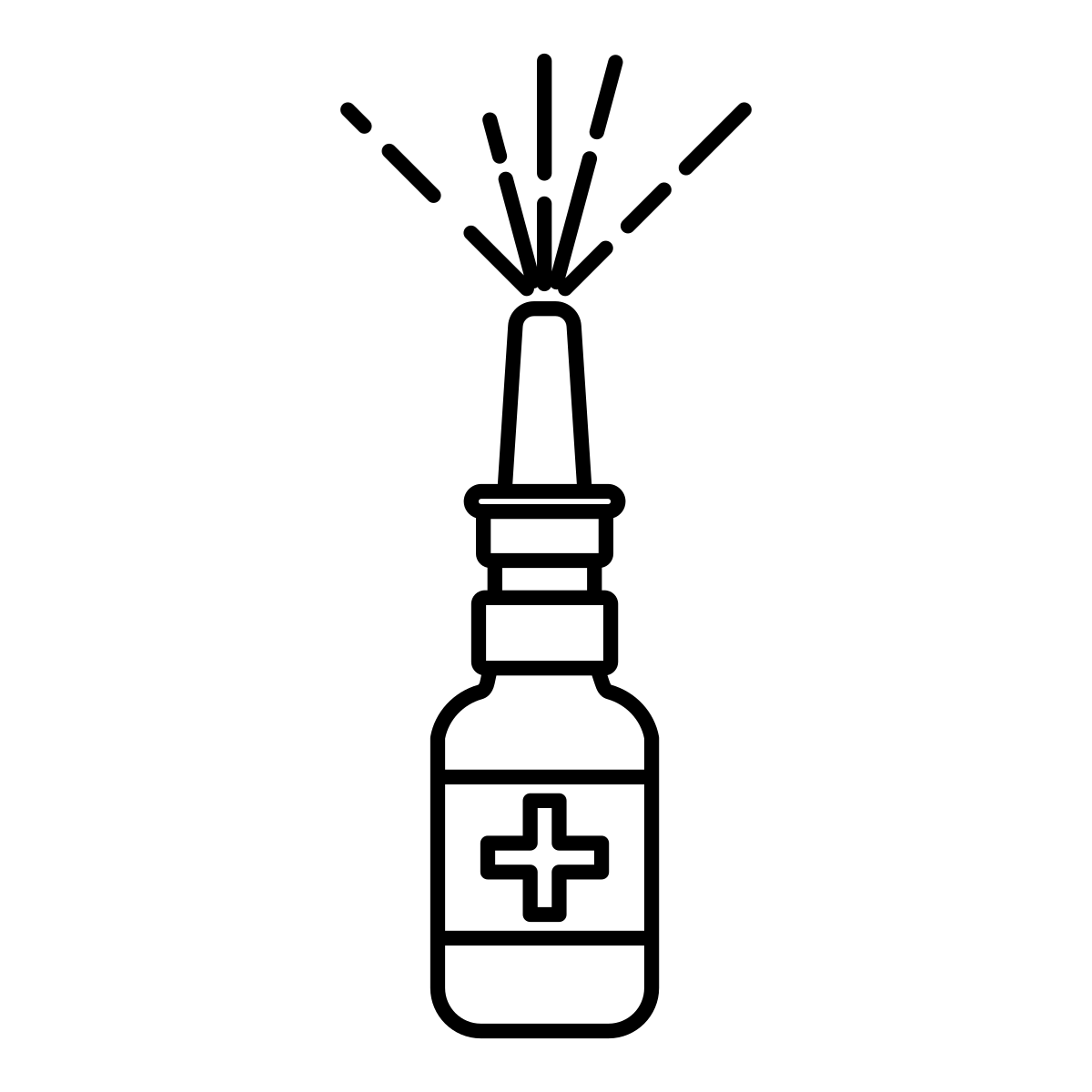
Mucosal immunization
Pneubera’s vaccine is administrated mucosally as a nasal spray. It builds a strong protection in the nasal mucosa and hinder the bacteria already at the point-of-entry. It also induce a strong systemic protection as a second line of defense.

No transmission
The vaccine is hindering bacterial colonization in the mucosa. Without colonisation, there is no bacteria in the nasopharynx, hence there is no transmission of bacteria to others. Currently, high colonisation in children strongly drive the disease prevalence in elderly due to transmission.

Universal protection
Pneubera’s vaccine is the first in a new class of pneumococcal vaccines building highly conserved antigens. The vaccine aims to protect against all serotypes of the bacteria. Traditional vaccine only covers certain serotypes; 15-23 out of 97 known serotypes.
PNEUBERA'S PNEUMOCOCCAL VACCINE
First in class.
Full coverage.
No transmission.

The challenge
Pneumococcal infections cause 1.5 million deaths per year
High unmet medical need
Pneumococcal infections cause otitis media, sinusitis, pneumonia and, when getting invasive, severe pneumonia, meningitis and sepsis. Over 1.5 million people die each year due to pneumococcal infections, and approximately 800,000 of them are children under 5 years. The infections cause 30+ millions of hospitalization cases in elderly each year.
High-cost serotype specific vaccines
Existing vaccines are all serotype specific, covering 15-23 out of 97 know serotypes of the pneumococcal bacteria. To broaden coverages, serotypes has been added but this cannot solve the long term problem, since we are reaching the limit of adding serotypes due to conjugate chemistry limitations; diluted antibody titers that increase slip-through cases; and increased manufacturing complexity and cost.
Serotype replacement
Prevalence of pneumococcal infections are continuous high, despite existing vaccines due to s/c serotype replacement. Vaccination with serotype-specific vaccines, PCVs, decrease the prevalence of serotypes included in the PCV but increase prevalence of non-included serotypes keeping incidence levels high. Emerging non-vaccine serotypes can be highly invasive and demonstrate antibiotic resistance. A broadly protective vaccine is needed to hinder continues serotype replacement.
Asymptomatic transmission
All existing vaccines are administrated intramuscularly eliciting limited mucosal protection focusing on battle the bacteria when already in blood stream with risk of causing invasive disease. To hinder disease development, both upper respiratory disease and invasive disease, as well as transmission, a mucosal protection hinder colonization is desirable.
The solution
New Broadly Protective Mucosal Vaccine
Pneubera is developing a novel pneumococcal vaccine candidate – targeting all serotypes and administered mucosally as a nasal spray.
New class of vaccines based on novel vaccine vector
The vaccine is based on Outer Membrane Vesicles (OMVs), decorated with multiple conserved pneumococcal antigens (antigens presented among all variants). OMVs are highly immunogenic, and no further adjuvant is needed. OMVs are well proven technology, e.g. used in licensed meningococcal vaccine, but is now developed to the next generation with the antigen decoration platform.
Universal vaccine protecting against all serotypes, hinder serotype replacement
Serotype prevalence differs extensively from region to region. A universal vaccine is effective worldwide and hinders serotype replacement problems, keeping the vaccine efficacy over time.
The vaccine’s antigens are detected by antibodies in sera from vaccinated animals for all (50+) tested serotypes, both PCV serotypes and non-PCV serotypes.
Mucosal vaccine inducing strong protection
The vaccine works upstream of PCV vaccines to prevent colonisation. Protects against colonisation of bacteria in the nose, hence protects against infection and transmission. The vaccine induces strong local IgA and Th17 response and significant systemic protection with high IgG titers.
Cost effective to produce
The production is based on traditional bacterial fermentation and a streamlined process. The cost of goods is estimated to just a fraction of the price of current PCVs.
Ready for clinic
The vaccine is now ready to enter clinical Phase I. An extensive pre-clinical package is developed in collaboration with Radboud University Medical Centre in the Netherlands. An approved clean GLP-tox study and an upscaled production process are already in place.
First in class, best in class
Based on novel OMV vaccine platform
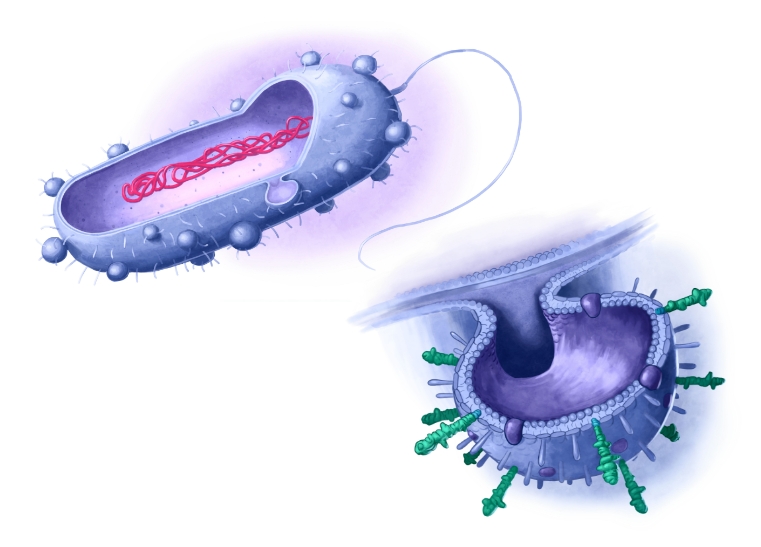
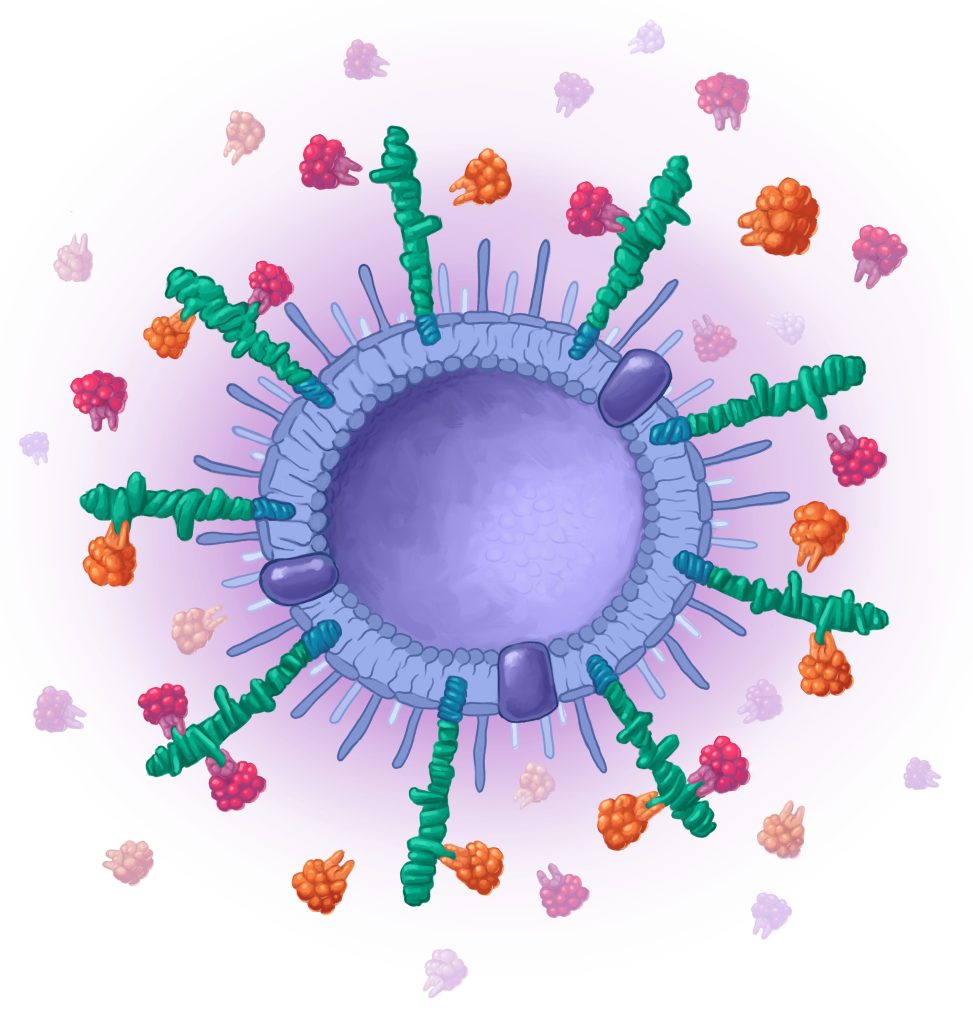
Vaccine candidate with OMVs displaying a large amount of multivalent antigens. The OMVs have immunostimulatory properties, hence no adjuvant needed.
